Functional groups
Other elements apart from hydrogen and carbon can be used to form different functional groups. Functional groups give the molecules their unique properties. Compounds with the same functional group behave in similar ways during reactions and can be grouped according to the functional group in place. It is the functional groups that react during chemical reactions.
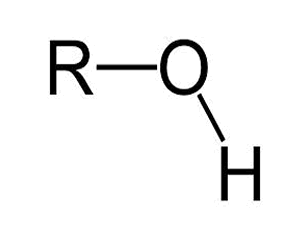
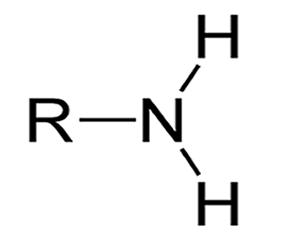
Compounds in which the principal functional group is the amino functional group (–NH2 ) are called amines and so ‘amine’ should appear in the systematic name.
The term ‘amino’ is used in compounds which contain –NH2 and a functional group of higher priority, e.g. such as in amino acids which contain the higher priority group –COOH.
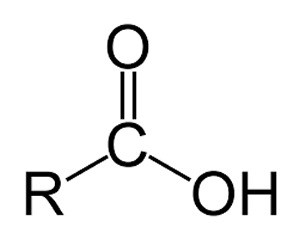
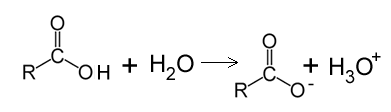
Organic acids contain the carboxyl group pictured on the left sometimes written as COOH. These molecules are weak acids that react with water to produce a hydronium ion.
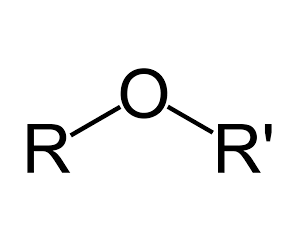
Molecules which have two carbons joined by an oxygen atom are known as ethers.
CH3CH2-O-CH2CH3
Diethyl ether, above, is the most commonly used ether. It is used as a solvent and as a general anaesthetic. Ether molecules belong to a very important class of biological molecules. Sugar molecules are linked together via ether links to each other to form long starch molecules and cellulose(in plants). Click to see an animation and more information on ether links.
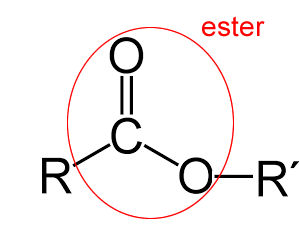
Esters are molecules with a unique fragrance and taste. Such molecules are formed via a reaction with an alcohol and an organic acid. These molecules are responsible for the natural smell and flavour of fruit.
Methyl butanoate has the smell of pineapple while pentyl ethanoate smells of bananas.
Click to see an animation of ester formation and the method of naming these molecules.
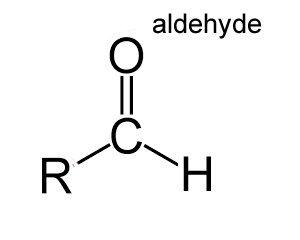
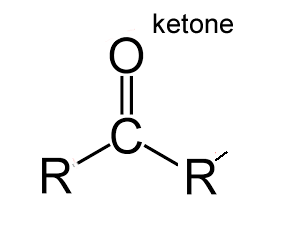
An aldehyde is an organic molecule that contains a carbonyl group (C=O), at the end of the molecule, with the central carbon bonded to a hydrogen and R group (R-CHO), as shown on the left.
Aldehydes differ from ketones in that the carbonyl is placed at the end of the carbon skeleton rather than between two carbon atoms of the backbone