Boiling, melting points and structure
A general trend, is that branched hydrocarbons have lower melting and boiling temperatures than their straight chain isomers.
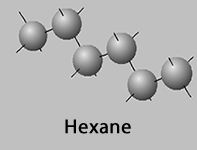
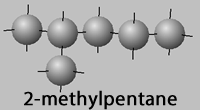
Lets look at hexane (C6H14) and its isomers, 2-methylpentane,
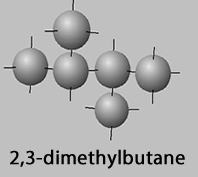
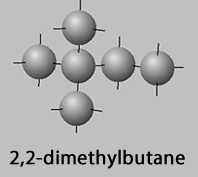
2,3-dimethylbutane, 2,2-dimethylbutane
| Melting temp (oC) | Boiling Temp ( oC) | |
 |
-95 | 69 |
 |
-153 | 61 |
 |
-129 | 58 |
 |
-100 | 50 |

Lets look at the trend of boiling and melting temperatures of branched molecules. As the side chains get closer together, the molecule becomes more spherical and this has an impact on the physical properties of the molecule such as its melting temperature and boiling temperature.
The more spherical the branched molecule becomes the lower its boiling temperature (BT), whereas the melting temperature (MT) increases.
Click on each compound to see the spherical nature
of each molecule.
Hexane
2-methylpentane,
2,3-dimethylbutane,
2,2-dimethylbutane
2,3-dimethylbutane and 2,2-dimethylbutane have the same side chains albeit separated by one carbon atom. Yet there is an 8oC difference in boiling point and a 29oC difference in melting point between the two molecules .
Explain the difference in boiling and melting temperatures. Refer to the space fill models.
To try and explain these trends let's look at the intermolecular forces at play between the alkane molecules involved. These forces are very weak and are known as London dispersion forces, commonly referred to as dispersion forces. These type of intermolecular forces occur between all molecules and atoms as a result of the motion of electrons that form instantaneous and short lived dipoles. Since these are the only type of forces acting between symmetrical molecules, a look into how molecular shape influences the impact these forces have on intermolecular forces of attraction.
Since dispersion forces are very weak they can only act over very small distances. Distance is the key to the impact dispersion forces have attracting molecules together.
Straight chain molecules have a higher melting temperature than their branched isomers due to the fact that they can pack more closely together in the solid crystal structure than branched chain molecules. For example take the packing of hexane molecules as shown on the right. These linear molecules can pack close together where the dispersion forces can exert a greater force of attraction. When one compares this tight packing of hexane with that of the isomer 2-methylpentane it is obvious that the molecules of 2-methypentane can not pack close to each other due to branching,
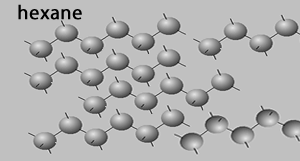
Straight chain molecules have a larger molecular surface area than their branched isomers so there is a larger area able to interact with other molecules in the liquid state resulting in higher boiling temperatures.
Consider the animation, on the right, of an interaction between two pentane molecules. As instantaneous dipoles form on the surface of one molecule a greater surface area enables a stronger interaction with the neighboring molecules, as shown on the right. This results in a greater force of attraction which requires a higher temperature to disrupt, hence a higher boiling temperature.
Now click to see an isomer of pentane, 2,2-dimethylpropane. This isomer of pentane is spherical and as such has a much smaller surface area than pentane and so has less interactions with neighboring molecules. This causes a lower BT for this isomer as compared to pentane.
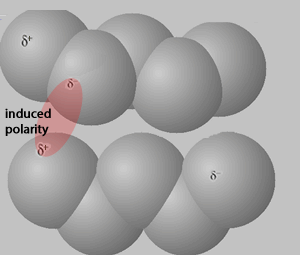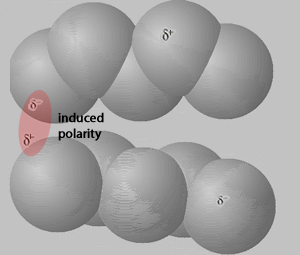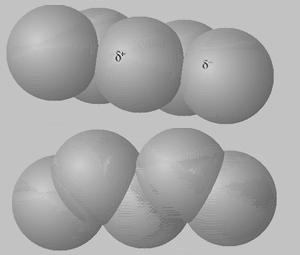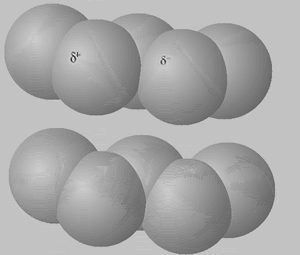
The animation above shows instantaneous dipoles forming on one molecule only, the top one, that results in induced dipoles on the neighboring molecule. In actual fact every molecule forms instantaneous dipoles that have an impact on neighboring molecules.
So in summary when comparing:
- straight chain molecules with branched molecules, straight chain molecules have a greater BT and MT than branched isomers of the same molecule.
With branched molecules
- the more branching the more spherical the molecules become and results in a higher melting point due to better stacking.
- the
more branching the more spherical the molecule becomes and results in lower boiling point due to a decrease in surface area.
The only exception to this trend is pentane and its isomer 2,2-dimethylpropane.
2,2-dimethylpropane is shown on the right and is a very symmetrical and more compact molecule than pentane. 2,2-dimethylpropane has a higher MT than pentane because it can form a tighter more highly packed crystal structure than the linear pentane molecule.
1) View the space fill structures of two isomers of octane.
a. Suggest which of the two molecules will have the highest BP and which one will have the highest MP.
b. Give a detailed explanation to justify your answer to a. above.
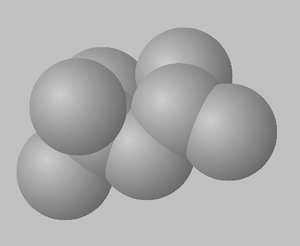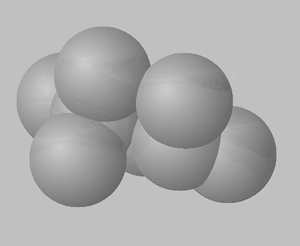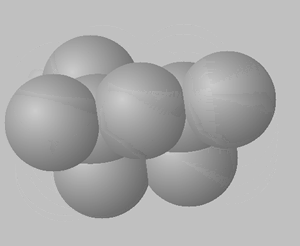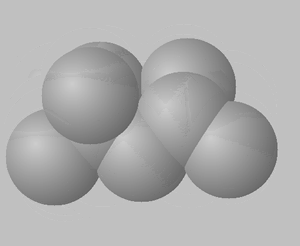
2,2,4-trimethylpentane
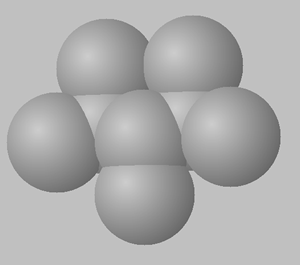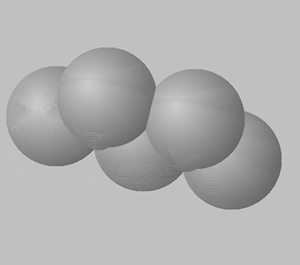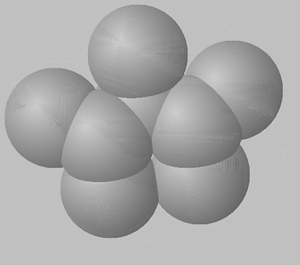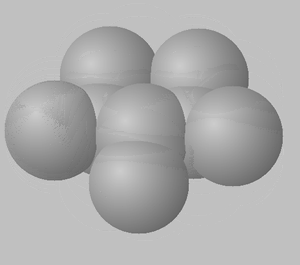
2,3,4-trimethylpentane