Electrolysis
in solutions exercises solution 3
Students should be aware that oxidation (the loss of electrons) occurs at the anode and reduction (the gain of electrons) occurs at the cathode.
3) An aqueous solution of
lead nitrate is electrolysed.
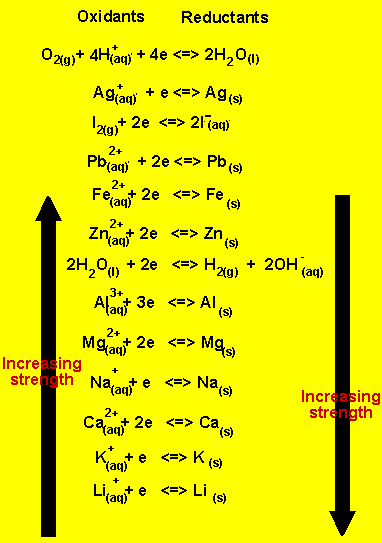
a) Using the table
on the right write the half equations that occur at the anode and the
cathode.
Since lead(II) ions are the strongest oxidant present they will
react at the cathode. At
the cathode(-) reduction of lead ions takes place according to the reaction
below.
Pb2+(aq)+ 2e => Pb(s)
At the anode(+) oxidation takes place. Water is the only reductant present and this will react according to the equation below.
2H2O(l) => O2(g) + 4H+(aq) + 4e
b) If the current through the electrodes is maintained for a long time
predict any changes in product formation.
Once the lead ions are fully deposited at the cathode water then becomes the strongest oxidant present and will react at the cathode according to the equation below.
2H2O(l) + 2e =>H2(g) + 2OH-(aq)