Electrolysis
in solutions exercises
1) An aqueous solution of
copper sulfate is electrolysed.
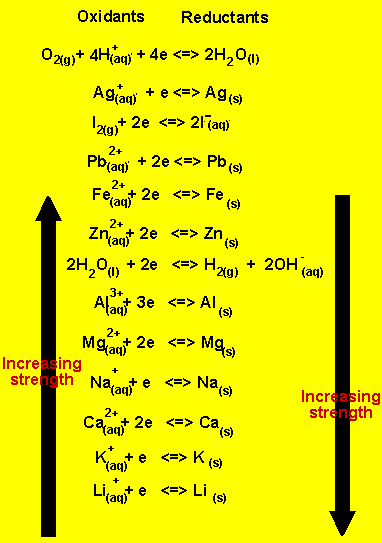
Cu2+(aq) + 2e => Cu(s))
appears on the table between Ag+ and Pb2+.
While SO-24 does not take part in the reaction.
a)What are the products formed
at the anode and at the cathode?
b) What happens to the pH of the solution?
Try four more similar exercises
2) A solution
of lead(II)iodide is electrolysed.
a) What are the products formed at the cathode and at the anode?
b) Give the reactions that
occur at the anode and at the cathode.
Use the reactivity series on the right.
3) An aqueous solution of
lead nitrate is electrolysed.
a) Using the table
on the right, write the half equations that occur at the anode and the
cathode.
b) If the current through the electrodes is maintained for a long time
predict any changes in product formation.
4) A solution containing
a mixture of lead nitrate, magnesium nitrate and silver nitrate was
electrolysed. The potential difference across the electrodes was maintained
for a long time until bubbles appeared at both electrodes.
a) Predict the products and
the order in which they are formed at the anode and the cathode.
b) Give equations for each
reaction that takes place.