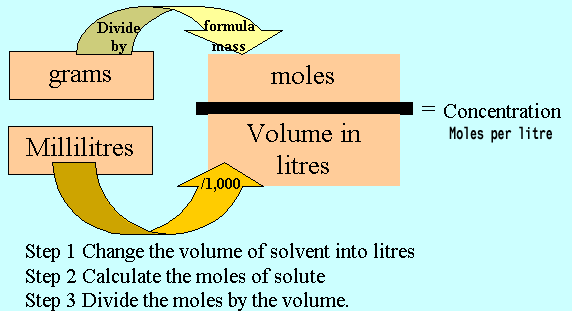
The concentration of moles per litre is known as Molarity (M)
Example
A Salt solution with a molarity of 2 has 2mole of salt in every litre
of solution.
A Sugar solution with a molarity of 0.2 has 0.2mole of sugar in every
litre of solution.
Example
4.4 grams of carbon dioxide were dissolved in 2 litres of water.
Calculate the molarity of the carbon dioxide solution formed.
Step 1 mole of carbon dioxide = 4.4 / 44 = 0.1
Step 2 concentration = 0.1 / 2 = 0.05M
Complete the table below. Click in the coloured squares
for extra information, double click to hide the information.
|