Solubility
saturated solution
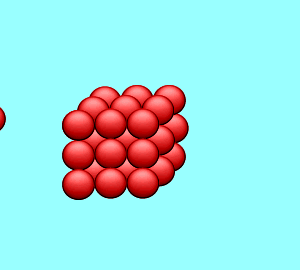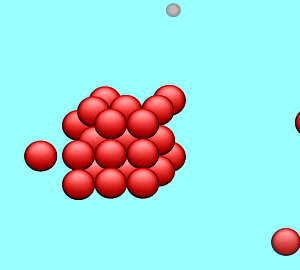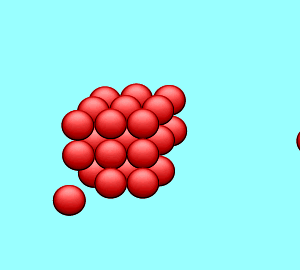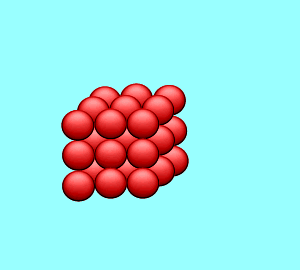
Solubility saturated solution |
||
| A solution is said to be saturated when no more solute can dissolve in the given amount of solvent at a particular temperature. If a solid crystal of the solute is placed in the saturated solution it will neither grow nor will it decrease in size. |
|
|
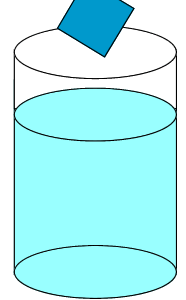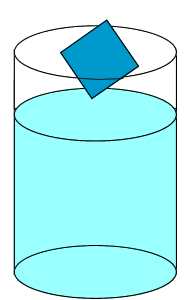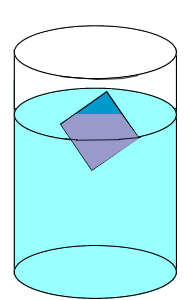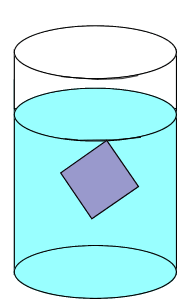
| The rate at which the solute particles stick to the crystal (precipitate) is the same as the rate at which the particles leave the crystal (dissolve). | 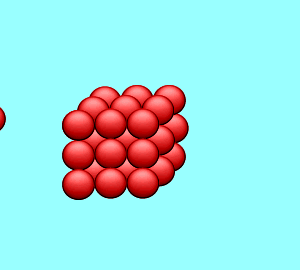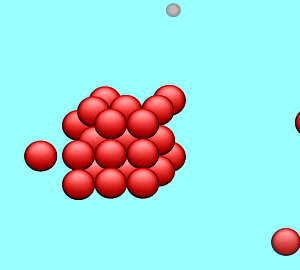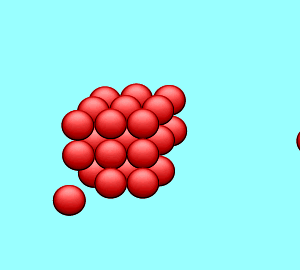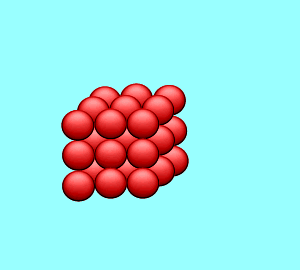 |
|