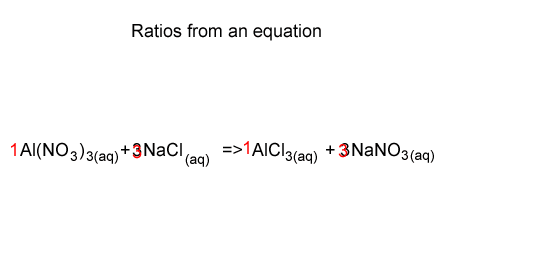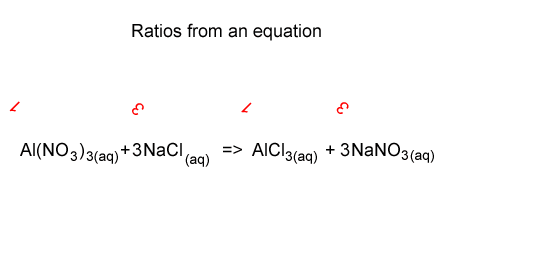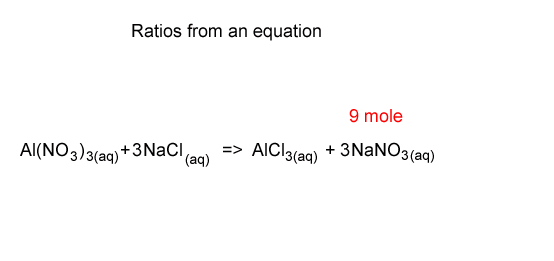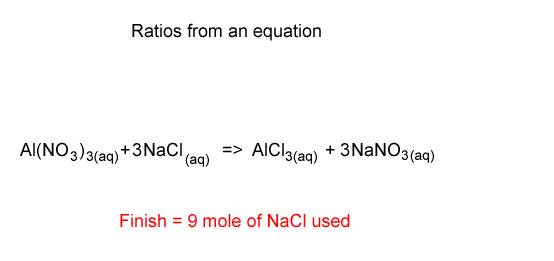Stoichiometry-using the ratios
Consider the question below.
If 9 mole of NaNO3 are formed how many mole of NaCl were used up?
Lets start with 9 mole of NaNO3 . Looking at the equation we see that the ratio of NaCl used and NaNO3 produced is 3 : 3 or 1 : 1. We therefore conclude that we used 9 mole of NaCl.