The dilution
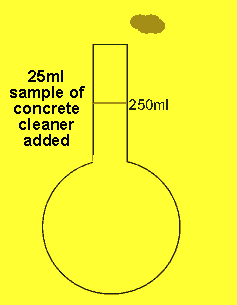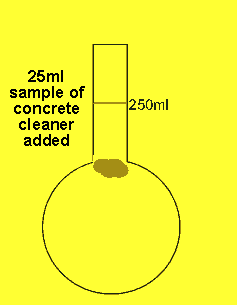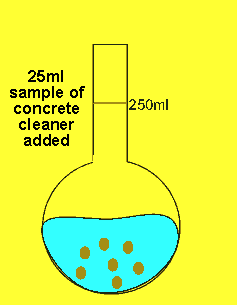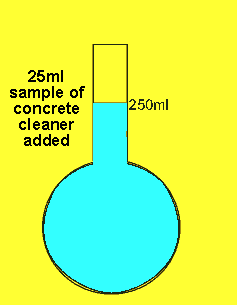
The 25ml sample of concrete cleaner is placed in the volumetric flask and distilled water added and made up to the mark
Click to hide this information
The dilution

The 25ml sample of concrete cleaner is placed in the volumetric flask and distilled water added and made up to the mark
Click to hide this information
The titration
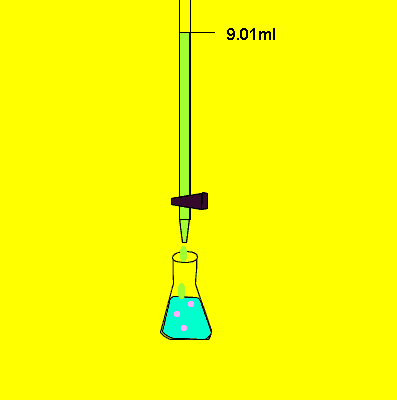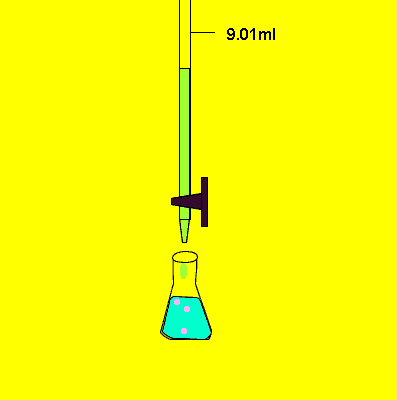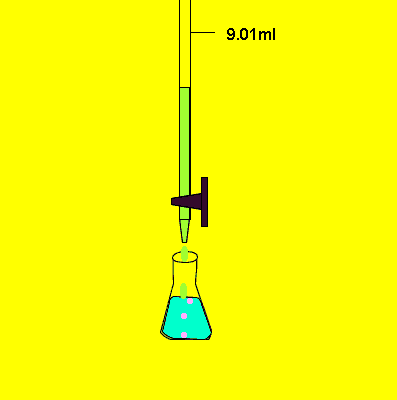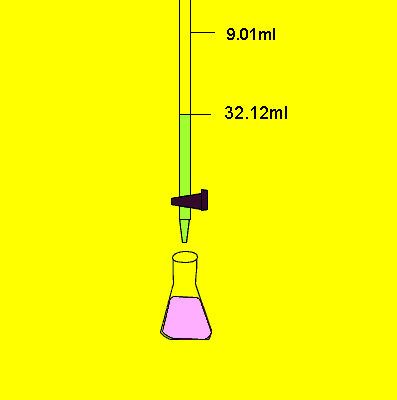
23.11mls of 0.2M sodium carbonatel solution is added to the 25ml sample before the indicator changed colour.
Click to hide this information
Taking a sample.
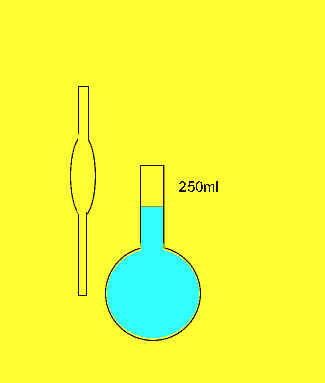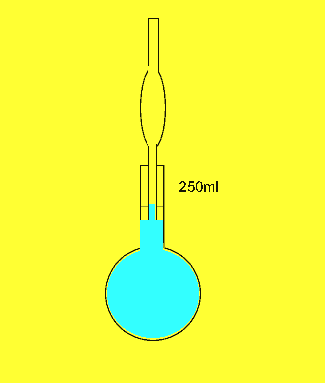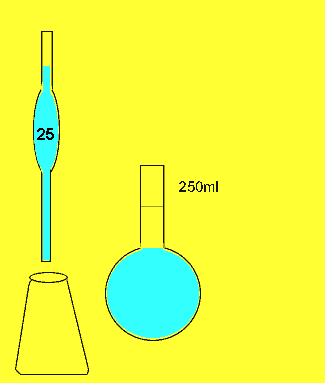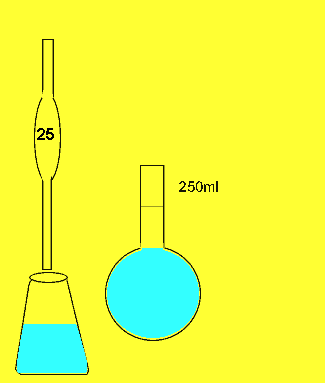
Pipettes are used to deliver accurate volumes of sample solutions.
Click to hide this information
Solution
Step 1 Calculate
the mole of sodium carbonate added
with the burette
Mole = volume X concentration
= 0.02311 X 0.2 = 0.0046
Step 2 Using the equation below calculate the number of mole of hydrochloric acid present required to react completely with 0.0046 mole sodium carbonate
![]()
0.0046 X 2 = 0.0092mole of hydrochloric acid in the 25ml sample.
Step 3 Find the mole of HCl in the original sample
(250/25) X 0.0092
=0.092 mole of HCl are present in the original 25ml sample of the concrete
cleaner.
Step 4 -Find the concentration of HCl in the original concrete cleaner.
mole of HCl /volume = Molarity = 0.092 /0.025 = 3.68M
Click to hide the solution
|
Volumetric analysis exercise 12 |
|||||||||
|
The concentration of hydrochloric acid in a liquid concrete cleaner was determined using the following method. A 25 ml sample was diluted to 250ml in a volumetric flask . 25mls of the resulting solution were pipetted into a conical flask and titrated against a 0.2M sodium carbonate solution. Sodium carbonate reacts with hydrochloric acid according to the equation below.
The results of the titration are shown on the right. What is the concentration of hydrochloric acid in the original sample?
Atomic mass Na =23, O =16, H=1, Cl=35.5, C =12 |
|
||||||||
|
Solution |
|||||||||