Experimental technique
Controlled experiments
Rate of reaction
Irene placed a 5 gram piece of calcium carbonate in a container of vinegar. The gas that evolved was trapped in a cylinder and its volume recorded. The time for the reaction to reach completion was also recorded.
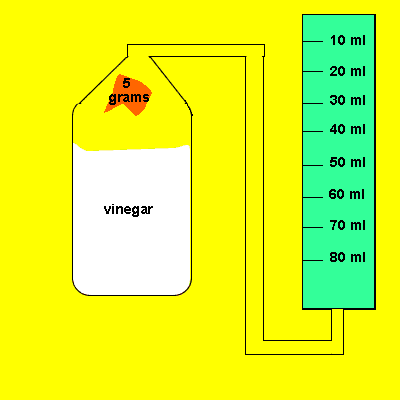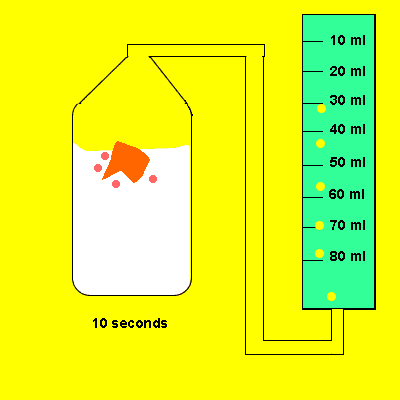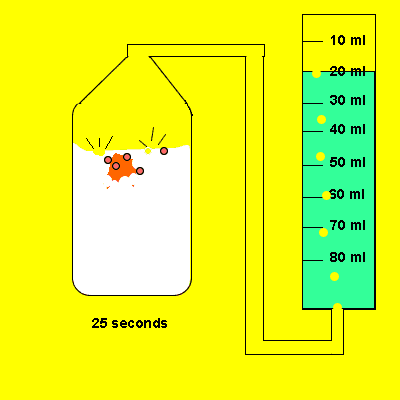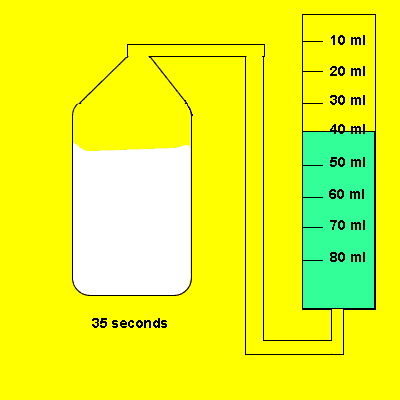
Irene later tried the same experiment with 5 grams of powdered calcium carbonate. The gas that evolved was trapped in a cylinder and its volume recorded. The time for the reaction to reach completion was also recorded.
Why is the same amount of gas produced each time?
What are the
variables that Irene must control when she repeats the experiment?
What was Irene trying to find?
What is her likely conclusion?
Solution
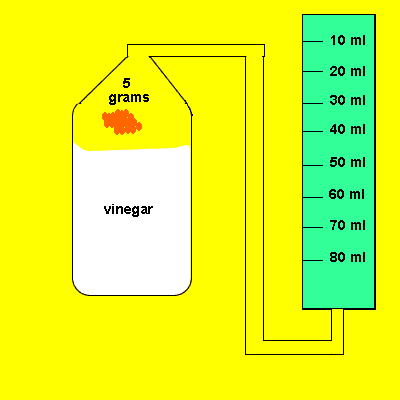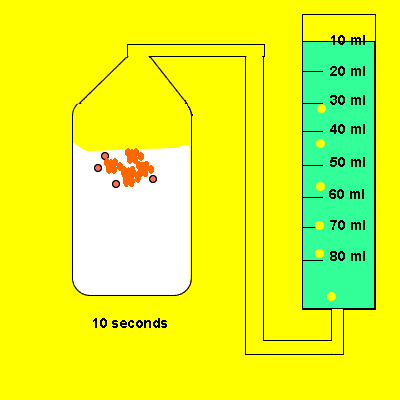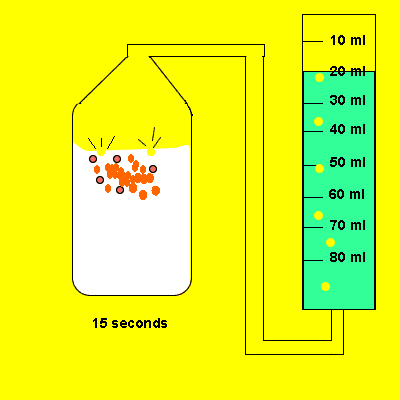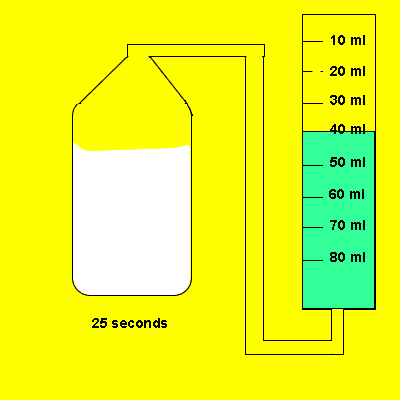
Design an experiment to find out if the temperature of the water influences the amount of gas dissolved in it.