Enzymes
Enzymes are proteins that act as catalysts:
- they participate in the reaction, but are not used up or altered during the reaction;
- do not affect the overall yield of product, only the rate at which the reaction proceeds, between 108 and 1010 faster than normal rates in absence of an enzyme.;
- specific for an individual reaction;
- reduce the activation energy by bringing the substrates into fixed positions that facilitates reaction.
All enzymes have a region on their surface where reactants attach, called the active site.
Enzymes work best within a narrow pH and temperature range. Heating the enzyme or altering the pH of the enzyme's environment can alter the shape of the active site and therefore render the enzyme inactive. The enzyme is said to be denatured. Denaturing occurs when the protein is forced to unravel and thus lose its unique shape.
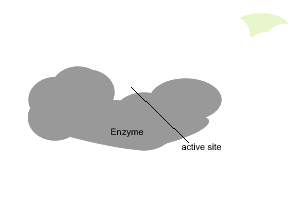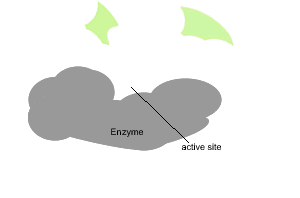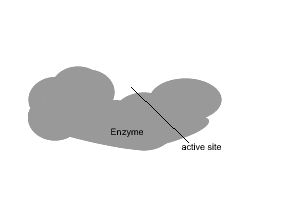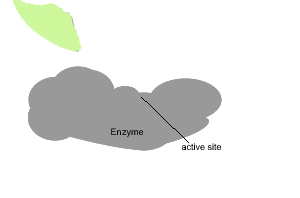
In the absence of enzymes most reactions proceed very slowly indeed. As shown on the right, in the absence of a catalyst the bonding of glucose molecules to form a branched polymer, glycogen, proceeds at a very slow rate. However the rate is increased in the presence of an organic catalyst (enzyme).
Enzymes lower the activation energy of a reaction.

Enzymes can be inhibited in a number of ways that fall into two categories, reversible and irreversible.
The first is competitive inhibition in which a molecule competes with the substrate molecules for binding on the active site and temporarily binds to the active site.
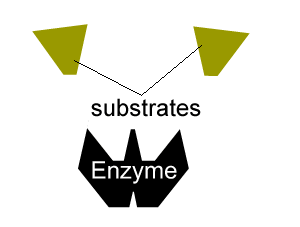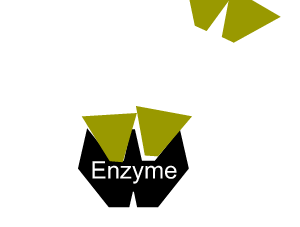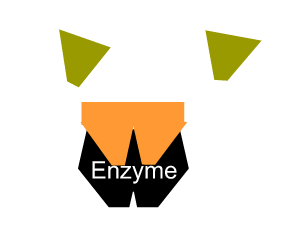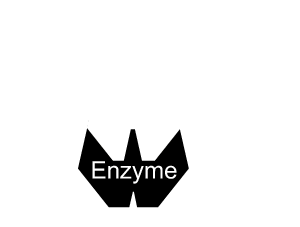
The second type of reversible inhibition is known as non-competitive inhibition. Here a molecule binds to the enzyme at a location other than the active site and either slows the rate of reaction, as shown on the right, or completely distorts the active site and renders the enzyme inactive.
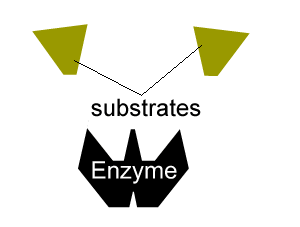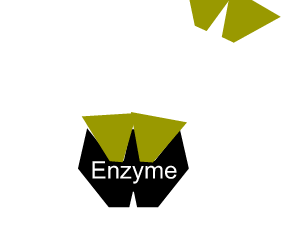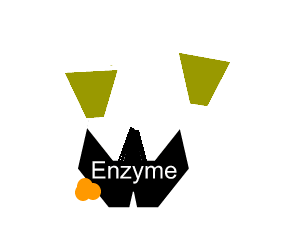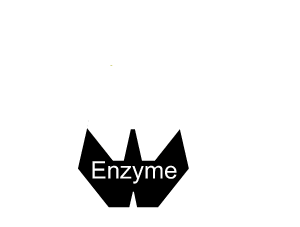
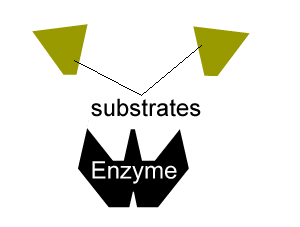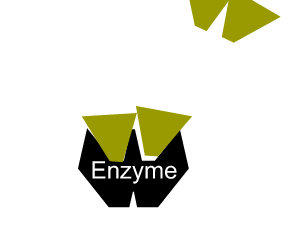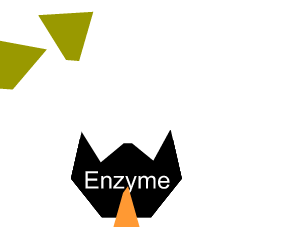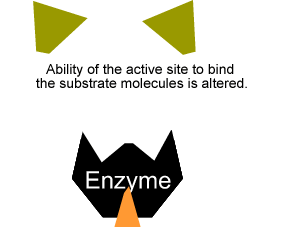
Inhibitors that bind to the enzyme and drastically alter the shape of the active site are known as allosteric inhibitors as pictured on the right.
Irreversible forms of inhibition include denaturing, which is mentioned above, and the irreversible binding of poisons to the enzyme. Binding of poisons, such as nerve gas, impairs the function of the enzyme.

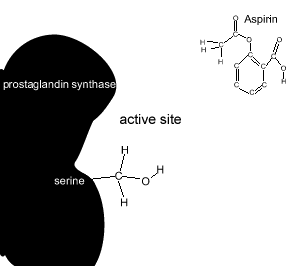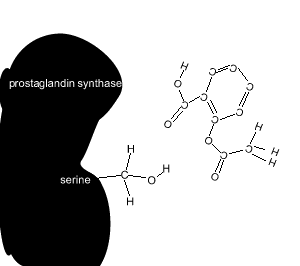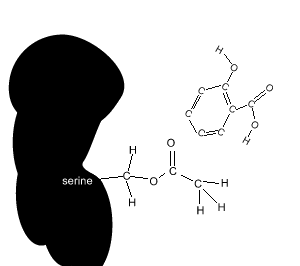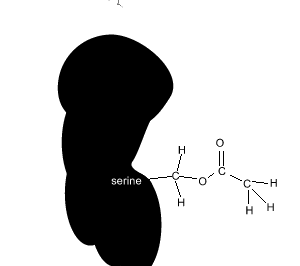
Aspirin reacts chemically with a serine amino acid in the active site. It covalently attaches an acetyl group to this amino acid and permanently changes the active site. Covalent bonds are strong and form permanent links.
Another medication called Ibuprofen also inhibits prostaglandin synthase. Unlike Aspirin, Ibuprofen is reversible. Reversible inhibitors are at equilibrium with the enzyme as shown by the equation below.
Enzyme + Ibuprofen <==> Enzyme-Ibuprofen complex.