Chemical reactions
Between atoms
We sometimes
have reactions between individual atoms. During such reactions atoms give,
take or share electrons in order to achieve a more stable electronic arrangement.
Click for more information
on stable electronic arrangements of atoms.
After exchanging electrons
atoms come together and form strong bonds between them known as chemical
bonds.
Click for more information on atoms
exchanging electrons.
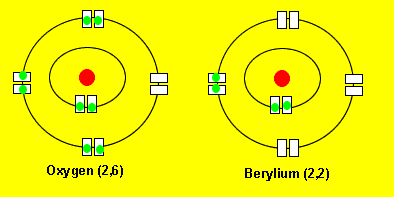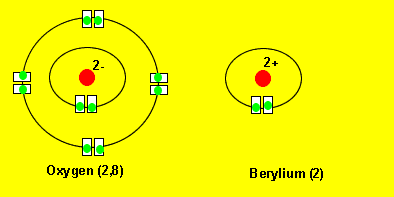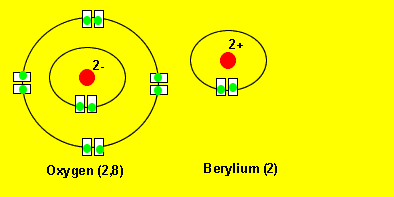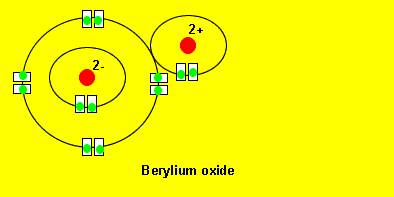
Oxygen reacts with berylium to form a new compound called beryllium oxide. Oxygen takes two electrons from beryllium to achieve a more stable electronic configuration. Notice how oxygen ends up having a 2,8 configuration and beryllium a 2 configuration. These type of configurations are very stable and you can see more detailed explanations by clicking on the links on the left.