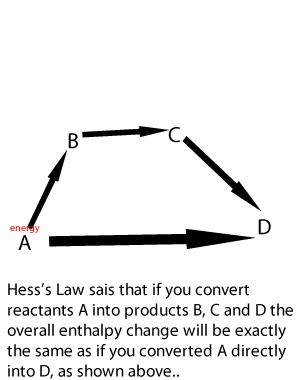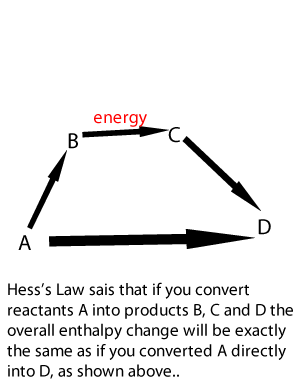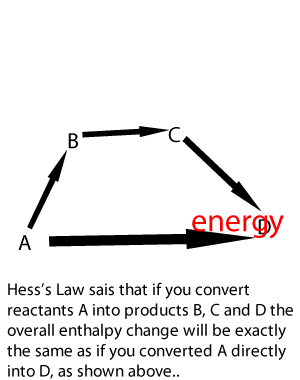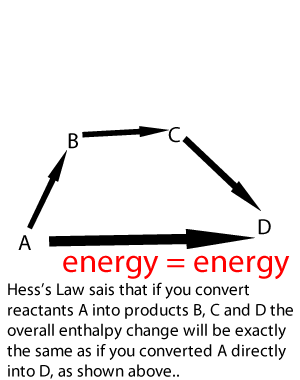
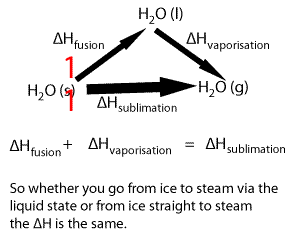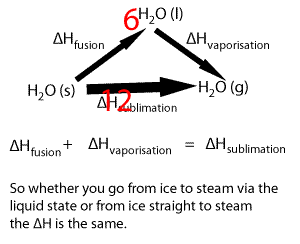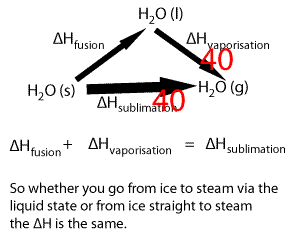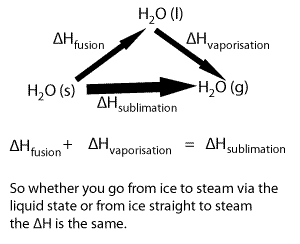
Lets take the following equations
1) ---- H2O(s) => H2O(l) ΔH = 6.00 kJ/mol
2) ---- H2O(l) => H2O(g) ΔH = 44.01 kJ/mol
Adding 1) and 2) we get
H2O(s) + H2O(l) => H2O(l) + H2O(g) ΔH = 50.01 kJ/mol
Now since H2O(l) appears on both sides of the equation we can cancel it out to get the equation below
H2O(s) => H2O(g) ΔH = 50.01 kJ/mol
Lets try some examples. But before we do remember:
- if you reverse the reaction the sign of the ΔH must change,
- if you add two reactions you add their ΔH,
- if you multiply an equation you must also multiply the ΔH.
Example 1
Find the ΔH of the reaction below CO 2(g) + H 2(g) => CO(g) + H 2O(g)
given the two equations below.
1) ----- 2H 2(g) + O 2(g) => 2H 2O(g) ΔH = - 484 kJ/mol
2) ----- 2CO(g) + O 2(g) =>2CO 2(g) ΔH = - 566 kJ/mol
Step 1 Reverse equation 2)
3) ----- 2CO 2(g) => 2CO(g) + O 2(g) ΔH = 566 kJ/mol
Step 2 Add equations 1) and 3) and dont forget to also add their ΔH
4) ----- 2CO 2(g) + 2H 2(g) => 2CO(g) + 2H 2O(g) ΔH = (566 - 484) = 82 kJ/mol
Step 3 Divide equation 4) by 2 and do the same to its ΔH
CO 2(g) + H 2(g) => CO(g) + H 2O(g) ΔH = 82/2 = 41 kJ/mol
Example 2
Calculate the ΔH of the reaction 2S(s) + 3O 2(g) =>2SO3(g) given
1) ----- S(s) + O 2(g) => SO2(g) ΔH = - 297 kJ/mol
2) ----- 2SO3(g) =>O 2(g) + 2SO2 (g) ΔH = 196 kJ/mol
3) ----- O2(g) + 2SO2 => 2SO3(g) ΔH = - 196 kJ/mol
Step 3 Multiply equation 1) by 2 and don't forget to multiply the ΔH
4) ----- 2S(s) + 2O 2(g) => 2SO2(g) ΔH = - 594 kJ/mol
Step 4 Add equations 3) and 4) and cancel any substance that appears on both sides, as shown on the right. Also add their ΔH
3O2(g)+2S(s) + 2O 2(g) => 2SO3(g) ΔH = - 790 kJ/mol
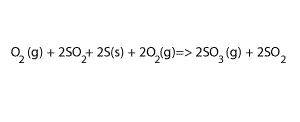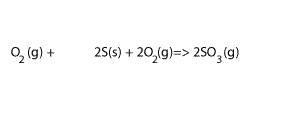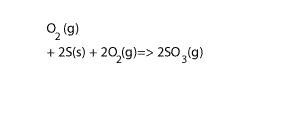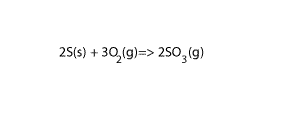
1) Calculate the ΔH of the direct synthesis of methanol according to the equation 2C(s) + O 2(g) + 4H2(g) => 2CH3OH (g) ΔH = ?
given the ΔH of the following reactions.
1) ----- 2CH3OH (g) + 3O 2(g) => 4H2O(g) + 2CO2 (g) ΔH = - 1352 kJ/mol
2) -----
C(s) + O 2(g) => CO2(g) ΔH = - 393 kJ/mol
3) ----- 2 H2(g) + O 2(g) => 2H2O(g) ΔH = - 484 kJ/mol
2) Calculate the ΔH of the following reaction 2CO(g) + O2(g) => 2CO2 (g) given
1) ----- C(s) + O 2(g) => CO2(g) ΔH = - 393 kJ/mol
2) ----- 2C(s) + O 2(g) => 2CO(g) ΔH = - 221 kJ/mol
3) Calculate the ΔH of the following reaction 2N2 (g) + 5O2(g) => 2N2O5 (g) given
1) ----- 2 H2(g) + O2 (g) => 2H2O(l) ΔH = - 571.6 kJ/mol
2) ----- N2O5 (g) + H2O(l) =>2HNO3(l) ΔH = - 76.6 kJ/mol
3) ----- N2 (g) + 3O2 (g) + H2 (g) =>2HNO3(l) ΔH = - 348.2 kJ/mol

4) Consider the exercise on the left.
Solution
Step 1
Step 2
Step 3
Continue with an activity